
Instant JChem
Create, explore and share chemical data
Summary
Chemical data management from your desktop
Instant JChem (IJC) is an out-of-the-box tool that allows scientists to create, manage and analyze chemical structures and related non-chemical data. It serves as a desktop application that gives a convenient and straightforward approach to the analysis of chemical and biological data. Instant JChem is a database-centric environment that supports query and sorting functionality, and handles large volumes of data (hundreds of thousands of structures), both in local and remote databases- without any additional administration. Its wide ranging functionality includes customizable database views, integration of library enumeration, and dynamic population of columns with singular and combined molecular property and descriptor predictions.


Book a demo

Download
Benefits
Why you need Instant JChem
Relational database management tool
Locally supported databases, easily managable schemas, trackable properties and simple sharing

View and search the content of your database
Customizable forms and tables, complex queries, easy export and visualization
Visualization and data analysis
Higlighting data trends with a variety of charts, conditional formatting and data pivoting feature
Customizability
Public API enables the removal of limitations on box products and allows the support of specific use cases.
Web-based extension
Plexus connect enables access to relational databases managed by Instant JChem in an online environment

Relational database management tool
Instant JChem - using our robust JChem Engine - can support a wide range of relational databases both locally (Derby is bundled) and remotely (Oracle, MySQL, PostgreSQL etc.). Schemas (database connections) can easily be created and administered, using the data of an existing data warehouse or importing your own data to populate it. (Read more and try data import). Chemical structures will be brought to a standard form, and you can add extra fields with various physical-chemical properties. All this data can be organized into projects, and shared with colleagues with different roles. All users can organize the data in custom views based on their own preference.
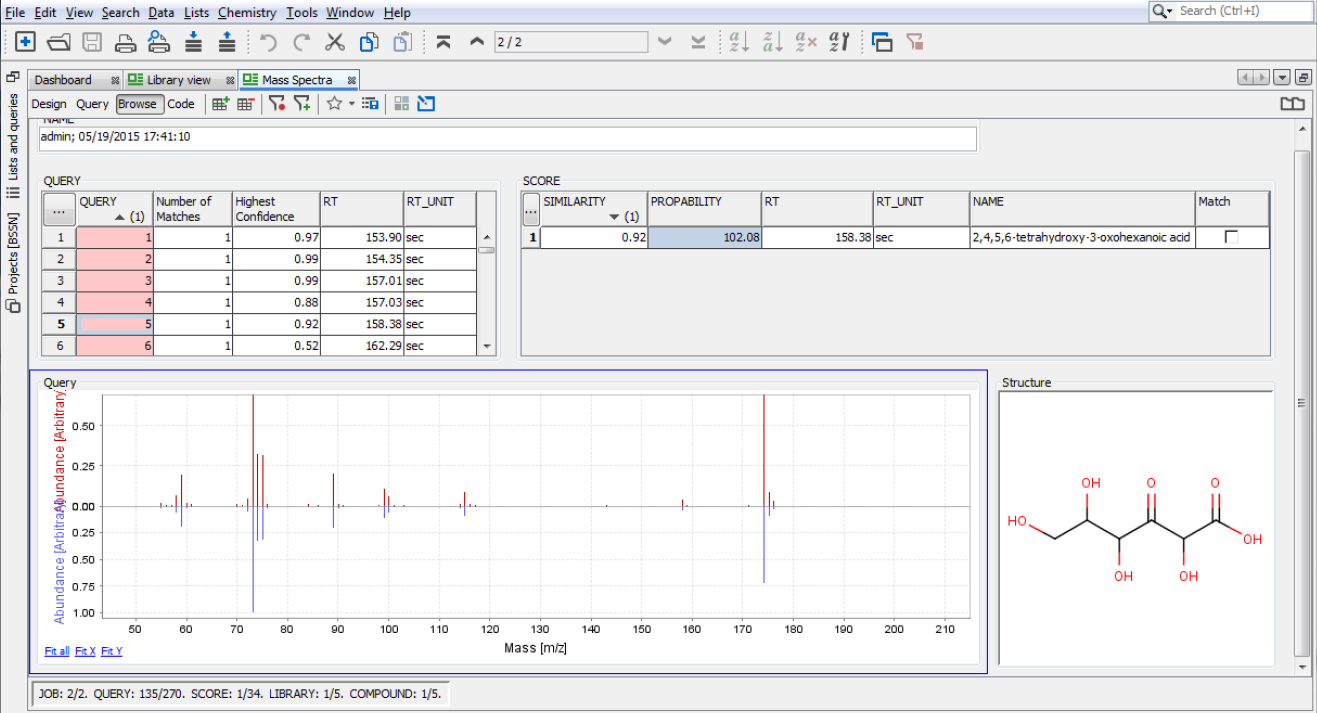
View and search the content of the database
The customizable forms and tables in Instant JChem allow users to exhibit and arrange the data based on personal needs. Data rows are easy to sort. Building even complex queries is straightforward in Instant JChem. Duplicate, substructure, full fragment, fragment and superstructure search methods are supported (more about querying). Queries and also selected compound lists can be saved for further use. Finally, exporting capabilities offer to print your results in SD file, MS Excel file, PDF file or any other text formats. The projects can be visualized in a web browser environment too, using our Plexus Connect.
Visualization and data analysis features
The data trends can be highlighted and visualized directly in Instant JChem, while it provides easy access to other tools for detailed data analysis.
- Use conditional formatting templates or create your own
- Display the values in four types of charts (histogram, scatterplot, radar chart and box plot)
- Use the data pivoting feature
Features
Customizability to suit your needs
Public API enables the removal of limitations on box products and allows the support of specific use cases.
- Use additional Java modules or create your own
- Custom scripts in Groovy for specific functionality
- MiniReg as custom solution for lightweight compound solution and assay handling
- Consultancy services available
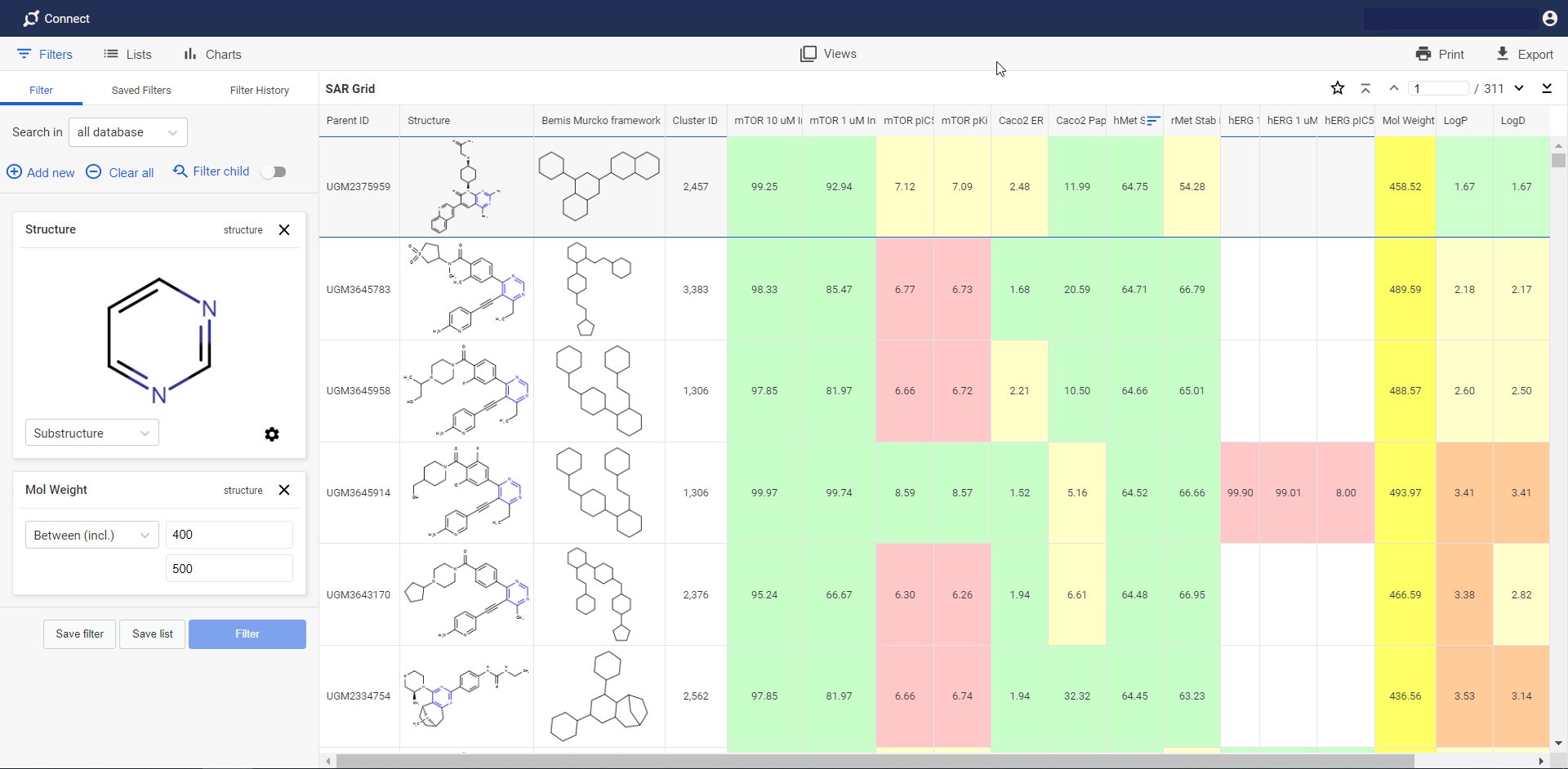
Plexus Connect
Web-based extension
- Plexus Connect is a powerful web-based extension of Instant JChem. It provides access to relational databases managed by Instant JChem in an online environment. Database setup, project management and user administration are through Instant JChem, making Plexus Connect an easy-to-operate tool for end users.
Knowledge Hub
Resources
Learn more about Instant JChem
Documentation
Related Products
Marvin
Full featured chemical editor for all platforms
Chemical Naming
Convert chemical names into structures
Markush Technology
Smart assistant for patent claim drafting and Markush analysis
Chemical Structure Representation
Standardization and correction of chemical structures
Chemicalize
Calculate properties instantly, search chemical data, and draw molecules online
Compound Registration
Normalize, check, validate and register chemical compounds
Reactor
High performance virtual synthesis engine
JChem for Office
Chemical structure handling, data analysis, visualization and reporting capabilities within MS Office
Design Hub
A single platform that connects scientific rationale, compound design and computational resources
JChem Engines
Search through tens of millions of chemical compounds and receive relevant query hits in seconds.
Calculators and Predictors
Execute high quality physico-chemical calculations and predictions.
Compliance Checker
Identify controlled substances with Compliance Checker and assign HS tariff codes with cHemTS - the easy way to comply with chemical regulations.
Discovery Tools
From clustering and diversity analysis for chemical libraries to 2D and 3D molecular screening
Instant JChem
Create, explore and share chemical data
ChemCurator
Computer-assisted chemical information extraction and analysis


